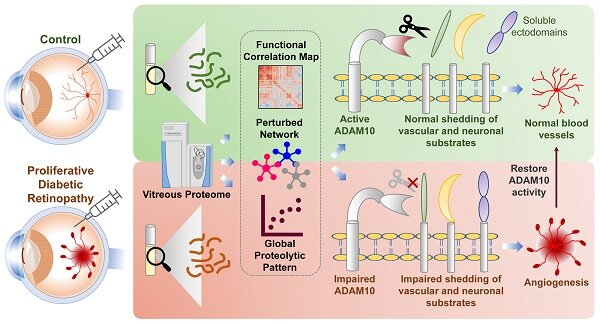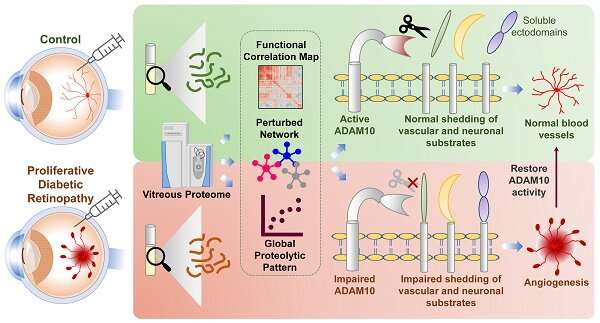
A recent study discovered a novel therapeutic target named ADAM10 that could be used to treat patients with Diabetic Retinopathy (DR), a condition that leads to blindness induced by prolonged diabetes. Abnormal blood vessel formation in the eyes of diabetic patients is a common phenomenon for DR, which could ultimately result in vision loss.
The study, published in the journal Theranostics, demonstrated that by restoring the function of ADAM10, a major shedding protein, it was possible in preclinical models to control the abnormal formation of blood vessels, offering an attractive therapeutic target to treat DR.
A collaborative effort involving researchers and clinicians from A*STAR’s Institute of Molecular and Cell Biology (IMCB), Duke-NUS Medical School, SingHealth, Singapore Eye Research Institute (SERI) and Singapore National Eye Centre (SNEC), the research team is exploring the potential of ADAM10 in various aspects of angiogenesis and how it may be translated into beneficial solutions for patients.
Currently, DR affects about 103 million people worldwide. According to a study done by the Centers for Disease Control and Prevention (CDC), DR is most common among diabetic patients, with almost one in three developing the condition. It is the leading cause of visual impairment and blindness in the working-age population globally.
DR usually presents without any symptoms in the early phase and is often diagnosed when the disease has advanced, requiring immediate treatment intervention. The current available form of treatment for DR is anti-VEGF (Vascular endothelial growth factor) injections, however only around half of DR patients respond to the treatment..
The research team, led by Dr. Jayantha Gunaratne, Senior Principal Investigator at IMCB, conducted a comprehensive analysis of the eye fluid samples from a well-defined cohort of proliferative DR patients against the control cohort to deduce impaired mechanistic aspects within the eye.
The findings show that eye fluids from DR patients displayed distinct protein patterns compared to the control cohort, implying that the molecular composition of eye fluids is reflective of the health status of the eye. By interrogating these altered profiles from DR patients, the team discovered impaired protein shedding by ADAM10 as a prominent disease feature of DR.
Working with the research team from Duke-NUS, these results were further validated using well-established cell-line and preclinical models with eye diseases through molecular, cell biological and functional assays to confirm the efficacy of the new target ADAM10 in controlling abnormal growth of blood vessels in the eye. With ADAM10 regulating various functional processes including neural and vascular aspects, it presents itself as an attractive therapeutic option for retinal angiogenic diseases.
This discovery provides key insights to the cause of DR and opens up a new path for developing effective therapeutics for DR patients, including patients who do not respond well to anti-VEGF treatments. Researchers also uncovered the involvement of other unknown potential molecular players in DR and the importance of understanding their mechanistic roles to effectively control or stop abnormal blood vessel formation in the eyes of DR patients.
Professor Hong Wanjin, Executive Director at A*STAR’s IMCB, said, “Through our collaborations with the local health care ecosystem, we have made significant progress with the discovery of therapeutic target ADAM10—This is a breakthrough for the scientific community and will help advance the development of targeted therapeutics leading to better health care outcomes.”
Dr. Jayantha Gunaratne, Senior Principal Investigator at A*STAR’s IMCB and lead author of the study, said, “This proteomics-centric discovery is a paradigm shift from conventional to non-conventional drug target identification, focusing on protein shedding activities of cell membrane proteins. It is a novel direction with immense potential for investigating effective therapeutics for several other diseases as well.”
Source: Read Full Article