According to data from Statistics Korea, cancer accounted for the majority of deaths in Korea in 2021, constituting 26% of all recorded cases. Similarly, cancer was the primary cause of death in 2020. Among the various types of cancer, lung cancer stands out as the most prevalent, posing significant challenges in terms of treatment due to its tendency to develop mutations that resists medication.
In drug screening research, such as genetic modification and targeted therapy, scientists widely employ organoids that possess the genetic characteristics of individual patients. However, replicating the intricate internal environment of a lung cancer patient’s body ,including the presence of tumors, solely using organoids, remains a formidable task.
Recently, a group of researchers including Professor Jinah Jang (Department of Mechanical Engineering and Department of Convergence IT Engineering) and Ph.D. candidate Yoo-mi Choi from the Department of Convergence IT Engineering at Pohang University of Science and Technology (POSTECH), along with Jinguen Rheey, CEO, and Haram Lee, principal researcher at Gradiant Bioconvergence achieved a significant breakthrough.
They successfully recreated the internal environment of a lung cancer patient using a hydrogel derived from decellularized extracellular matrix (LudECM) obtained from porcine lungs. The research findings were published in Biofabrication, the international journal.
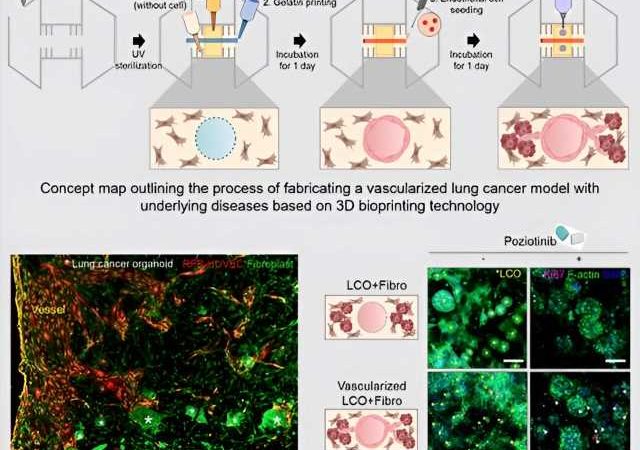
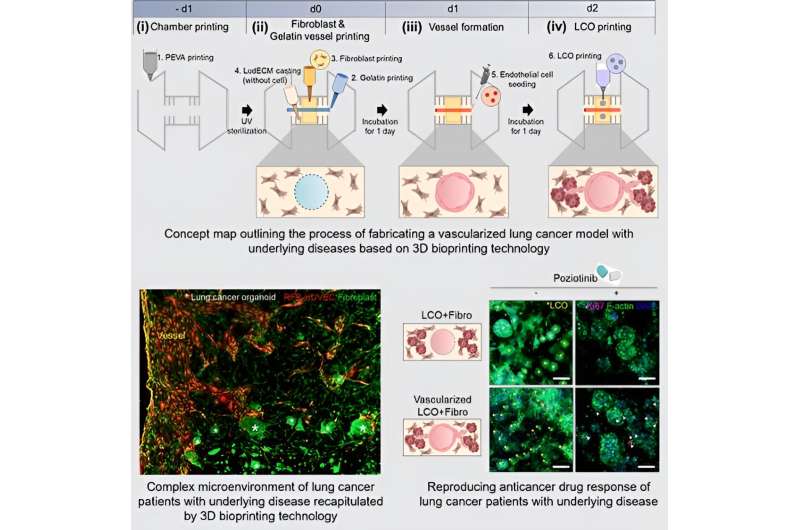
The research team developed three distinct types of bioinks utilizing organoids derived from lung cancer patients, fibroblasts sourced from patients with an underlying disease (fibrotic lung), and vascular cells. They employed LudECM as a material to replicate the intricate tumor microenvironment (TME) consisting of organoids, stromal cells, and vascular cells.
Cultivating lung cancer organoids in LudECM successfully preserved the specific lung cancer subtype and genetic mutation characteristics of the patients. Notably, LudECM exhibited significantly heightened sensitivity compared to conventional Matrigel when testing drug reactions in lung cancer organoids combined with fibrosis models.
In addition, the research team utilized 3D bioprinting technology to create a lung cancer model featuring perfusable vessels and fibrotic lung environment. Consequently, they achieved the successful recreation of the internal environment found within the bodies of lung cancer patients with underlying diseases, now in a laboratory setting. The generated lung cancer model remarkably replicated the complex surroundings of actual tumors, thereby enabling more precise drug evaluations than previously possible.
Through experiments conducted with clinical drugs, the research team observed that a lung cancer model with fibrosis has increased drug resistance compared to a general lung cancer model. This finding underscores the potential impact of fibrotic lung on the effectiveness of anti-cancer medications.
Furthermore, the team’s vascularized model demonstrated instances where drug delivery could be impeded due to drug absorption into surrounding matrices or interactions between cancer cells and stromal cells. Consequently, utilizing the research team’s model holds promise for providing personalized treatment options tailored specifically to lung cancer patients with underlying disease.
Professor Jinah Jang who led the research explained, “Choosing appropriate anti-cancer drugs presents a challenge for lung cancer patients, given the presence of complications and associated risk factors.” She continued, “I’m confident that this research will pave the way for customized treatments targeting other solid cancers that are accompanied by fibrosis.”
The research team’s LudECM, acknowledged as a valuable biomaterial for culturing lung organoids, has garnered recognition. The technology employed for this purpose has been patented and transferred to EDmicBio, currently undergoing commercialization processes.
More information:
Yoo-mi Choi et al, 3D bioprinted vascularized lung cancer organoid models with underlying disease capable of more precise drug evaluation, Biofabrication (2023). DOI: 10.1088/1758-5090/acd95f
Journal information:
Biofabrication
Source: Read Full Article