Major fungal infections have become more common across the globe, and one unexpected phenomenon among the rise of fungi is life-threatening infections as a result of a complication of certain immunotherapies and small molecule kinase inhibitors.
A scientist at the Hackensack Meridian Center for Discovery and Innovation (CDI) has identified the specific mechanistic cause of one such phenomenon, which will likely save lives into the future, via a new publication.
The paper “C5a-licensed phagocytes drive sterilizing immunity during systemic fungal infection” appeared in the journal Cell on May 22.
“Our findings will assist clinicians in their understanding of how these life-threatening infections are emerging,” said Jigar Desai, Ph.D., assistant member of the CDI, assistant professor of medical sciences at the Hackensack Meridian School of Medicine, and first author of the paper. “These findings may help doctors and scientists alike better understand how some of these cases arise—and how to avoid them.”
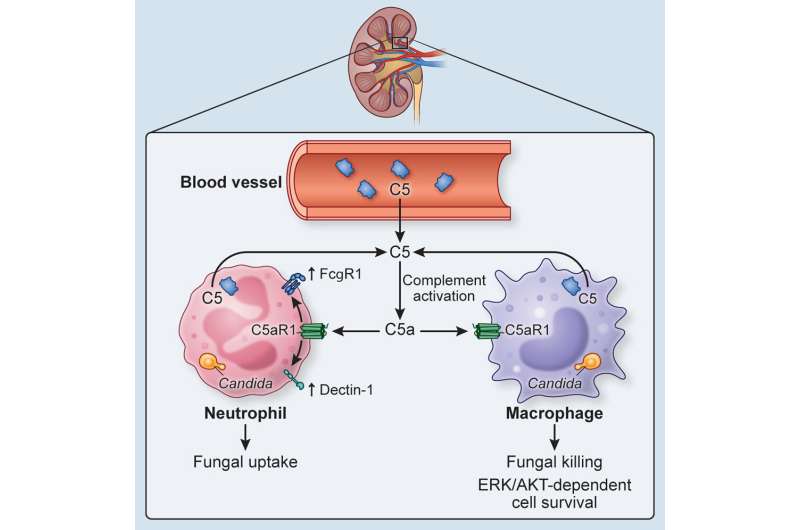
The team of scientists established that the C5a protein, the penultimate effector constituent of the complement pathway, is key to the body’s innate ability to fight systemic fungal infections. Additionally, the team also identified enhanced complement pathway signature acts as a predictive biomarker for systemic candidiasis. With the use of animal models, patient data and sera, the team showed how C5a and its downstream effects are crucial for the body’s immune cells, specifically neutrophils and macrophages, to clear the fungus Candida albicans, when it has overtaken the body’s natural defenses.
Desai and the team—which includes colleagues from the National Institutes of Health, Duke University, and Mount Sinai, among others—showed this in stages, both in animal models and in patient serum, by isolating what roles the C5 plays.
In addition to uncovering induced complement signature as a potential biomarker for systemic candidiasis, this work will be highly impactful in the clinical setting, where complement C5-targeted therapeutics, such as the anti-C5 monoclonal antibodies eculizumab/ravulizumab (as well as the C5a receptor inhibitor, avacopan) are the treatment of choice. In these settings, findings from this work emphasizes the importance of vigilant surveillance for opportunistic fungal infections, where early diagnosis can improve patient outcomes.
“Our findings establish a new paradigm in immunobiology, demonstrating for the first time the direct critical role of cell-intrinsic complement generation for effective host defense against Candida,” write the authors. “The multifaceted translation of our work shows promise for the development of individualized risk stratification and prognostication strategies in patients at-risk for invasive fungal disease.”
Desai, a fungal expert who joined the CDI last year, has had other recent publications.
In two papers in 2022 he and colleagues showed a particular genetic deficiency may weaken certain people to the onslaught of a certain plant pathogenic fungus, and also explore how systemic candidiasis may actually carry enhanced mortality following the use of broad-spectrum antibiotics. Those papers were published in The Journal of Clinical Investigation, and Cell Host and Microbe.
“Jigar Desai is uncovering novel insights into life-threatening fungal diseases,” said David Perlin, Ph.D., chief scientific officer and executive vice president of the CDI, who is also an expert in fungal infections. “As we know, this is an emerging health problem, and it’s key to have his work drive our understanding forward.”
More information:
Jigar V. Desai et al, C5a-licensed phagocytes drive sterilizing immunity during systemic fungal infection, Cell (2023). DOI: 10.1016/j.cell.2023.04.031
Journal information:
Cell Host and Microbe
,
Journal of Clinical Investigation
,
Cell
Source: Read Full Article
