A team of UK-based scientists has recently evaluated the effectiveness of coronavirus disease 2019 (COVID-19) vaccines in preventing household transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in England.
Their findings indicate that the vaccines effectively reduce transmission of SARS-CoV-2 alpha and delta variants from breakthrough infections in the household setting, despite having reduced efficacy in preventing infection. The study is currently available on the medRxiv* preprint server.
Background
The COVID-19 mass vaccination program was first introduced in the UK in December 2020 with the mRNA-based BNT162b2 vaccine (Pfizer/BioNTech) and adenoviral vector-based ChAdOx1 (Oxford/AstraZeneca) vaccine.
The primary aim of the vaccination program was to protect the most vulnerable population from infection and severe disease, including older adults, immunocompromised patients, and those with preexisting health conditions. In addition, healthcare workers and other frontline workers who are at higher risk of SARS-CoV-2 exposure were prioritized for the vaccination.
In the current study, the scientists have investigated the effectiveness of BNT162b2 and ChAdOx1 vaccines in preventing delta and alpha breakthrough infection and transmission among household contacts in England.
Study design
The study was conducted on individuals with primary infections (index cases) and their respective household contacts. The information on the vaccination status of participants was obtained from the National Immunization Management System, UK.
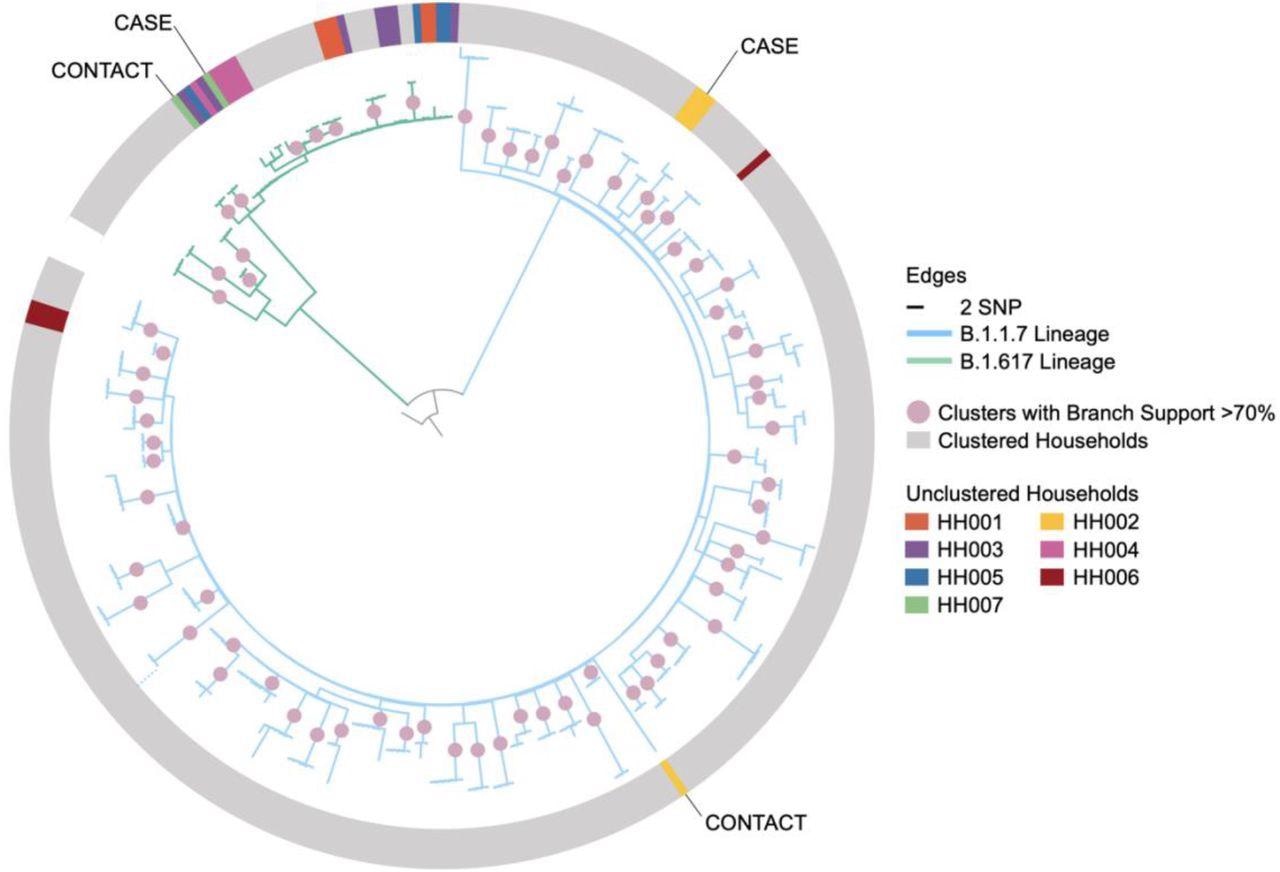
The participants (both index cases and contacts) were asked to collect respiratory samples on days 1, 3 and 7 post-recruitment using self-testing kits. The samples were tested later on by reverse transcription polymerase chain reaction (RT-PCR). A subset of samples was subjected to genomic sequencing to identify the alpha and delta variants.
Important observations
The study involved a total of 195 index cases and 278 contacts. On average, each household had 2 contacts. About 77% and 52% of index cases and contacts had received at least one vaccine dose at the time of recruitment, respectively. About 41% of contacts tested positive for SARS-CoV-2 within one week after recruitment.
Age- and variant-specific risk of infection
Of all index cases, 99 had the alpha infection, 24 had delta infection, and 52 had infections with unknown variants. In addition, 20 index cases did not test positive again after recruitment.
In the absence of vaccination of either case or contacts, the risk of delta infection transmission was 1.64-times higher than that of alpha infection transmission. Compared to children, adult contacts aged above 18 years were 1.19-times more susceptible to delta infection. The risk of viral transmission from delta index cases aged 50 years or above was comparatively higher than those aged 18 – 49 years.
Vaccine efficacy
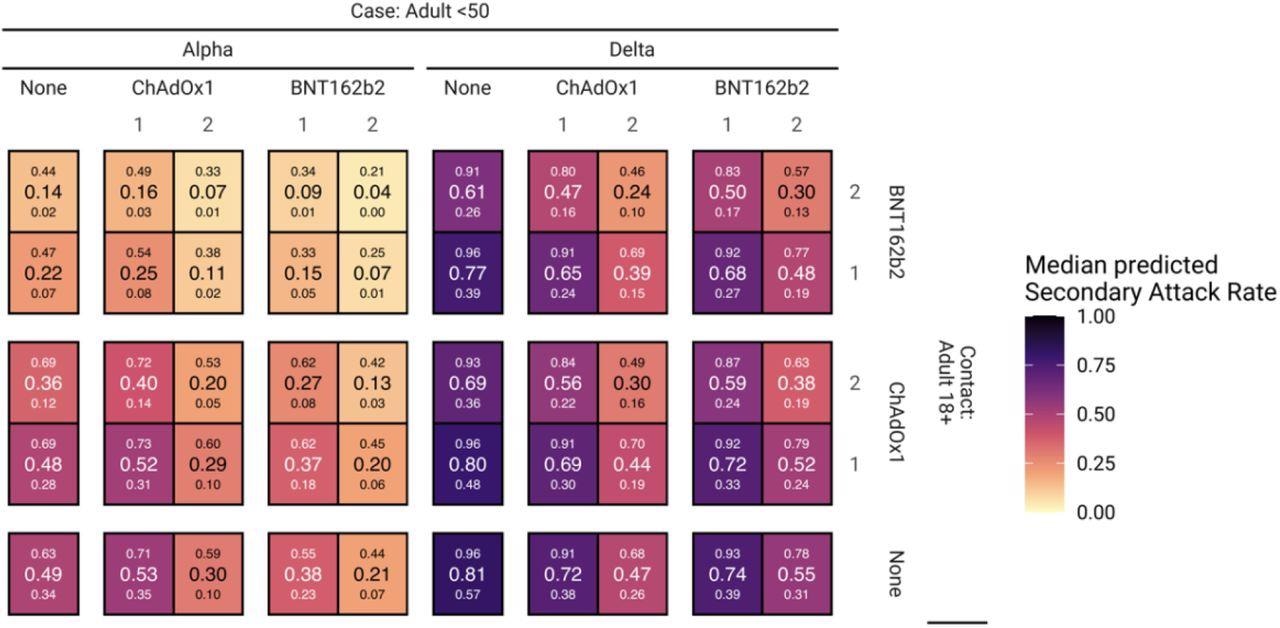
The effectiveness of two doses of BNT162b2 and ChAdOx1 vaccines against delta infection transmission was 31% and 42%, respective. The effectiveness was similar for alpha infection transmission.
Regarding protection against acquiring alpha and delta infections, the BNT162b2 vaccine had 71% and 24% effectiveness, respectively. For the ChAdOx1 vaccine, the effectiveness was 26% and 14% against the alpha and delta infections, respectively.
Prevalence of secondary infection
In unvaccinated households, the rates of secondary infections with alpha and delta variants among adults were 49% and 81%, respectively. The highest effectiveness of both vaccines against the secondary infection rate was observed when both cases and contacts were fully vaccinated.
Study significance
The study findings reveal that both mRNA-based and adenovirus vaccine vector COVID-19 vaccines effectively reduce the risk of viral transmission from persons who have developed alpha or delta infection despite being fully vaccinated. However, both vaccines have relatively lower efficacy in preventing delta infections.
The considerable level of protection against household SARS-CoV-2 transmission observed in the study highlights the importance of COVID-19 vaccines in limiting the risk of COVID-19, particularly in indoor setups where cases and contacts remain in close proximity for a prolonged period of time.
*Important Notice
medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
- Clifford S. 2021. Effectiveness of BNT162b2 and ChAdOx1 against SARS-CoV-2 household transmission – a prospective cohort study in England, medRxiv, https://www.medrxiv.org/content/10.1101/2021.11.24.21266401v2
Posted in: Child Health News | Men's Health News | Medical Research News | Women's Health News | Disease/Infection News
Tags: Adenovirus, Children, Coronavirus, Coronavirus Disease COVID-19, Efficacy, Genomic, Genomic Sequencing, Healthcare, Immunization, Phylogeny, Polymerase, Polymerase Chain Reaction, Respiratory, SARS, SARS-CoV-2, Severe Acute Respiratory, Severe Acute Respiratory Syndrome, Syndrome, Transcription, Vaccine

Written by
Dr. Sanchari Sinha Dutta
Dr. Sanchari Sinha Dutta is a science communicator who believes in spreading the power of science in every corner of the world. She has a Bachelor of Science (B.Sc.) degree and a Master's of Science (M.Sc.) in biology and human physiology. Following her Master's degree, Sanchari went on to study a Ph.D. in human physiology. She has authored more than 10 original research articles, all of which have been published in world renowned international journals.
Source: Read Full Article
