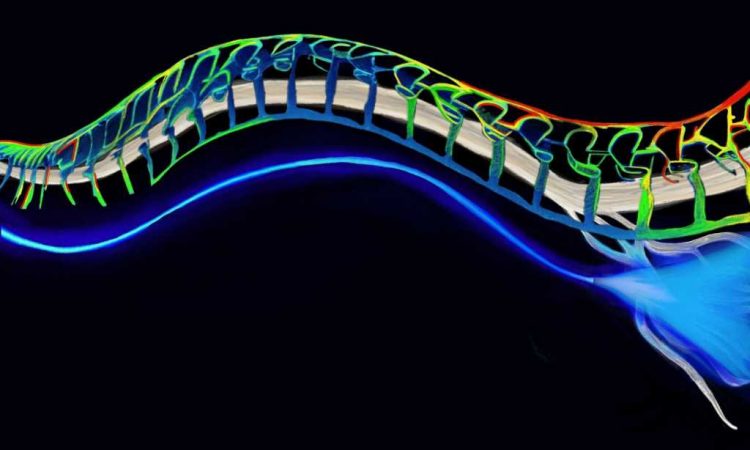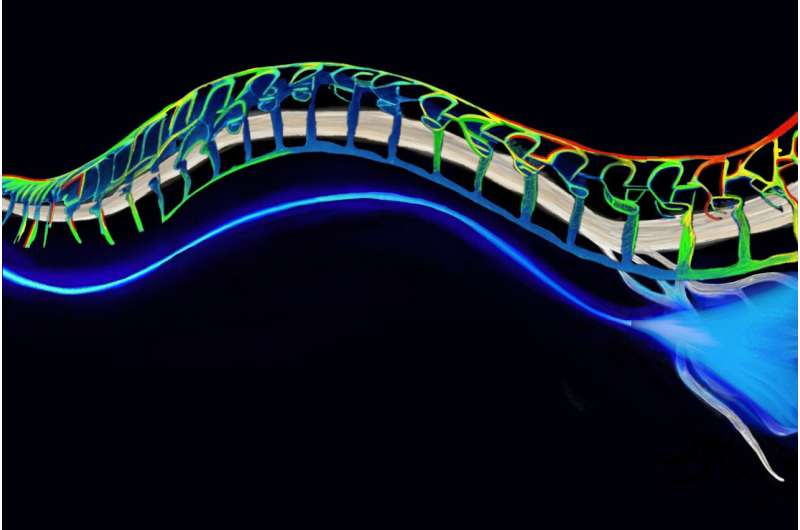
Engineers at MIT have developed soft and implantable fibers that can deliver light to major nerves through the body. When these nerves are genetically manipulated to respond to light, the fibers can send pulses of light to the nerves to inhibit pain. The optical fibers are flexible and stretch with the body.
The new fibers are meant as an experimental tool that can be used by scientists to explore the causes and potential treatments for peripheral nerve disorders in animal models. Peripheral nerve pain can occur when nerves outside the brain and spinal cord are damaged, resulting in tingling, numbness, and pain in affected limbs. Peripheral neuropathy is estimated to affect more than 20 million people in the United States.
“Current devices used to study nerve disorders are made of stiff materials that constrain movement, so that we can’t really study spinal cord injury and recovery if pain is involved,” says Siyuan Rao, assistant professor of biomedical engineering at the University of Massachusetts at Amherst, who carried out part of the work as a postdoc at MIT. “Our fibers can adapt to natural motion and do their work while not limiting the motion of the subject. That can give us more precise information.”
“Now, people have a tool to study the diseases related to the peripheral nervous system, in very dynamic, natural, and unconstrained conditions,” adds Xinyue Liu Ph.D. ’22, who is now an assistant professor at Michigan State University (MSU).
Details of their team’s new fibers are reported in a study appearing in Nature Methods. Rao’s and Liu’s MIT co-authors include Atharva Sahasrabudhe, a graduate student in chemistry; Xuanhe Zhao, professor of mechanical engineering and civil and environmental engineering; and Polina Anikeeva, professor of materials science and engineering, along with others at MSU, UMass-Amherst, Harvard Medical School, and the National Institutes of Health.
Beyond the brain
The new study grew out of the team’s desire to expand the use of optogenetics beyond the brain. Optogenetics is a technique by which nerves are genetically engineered to respond to light. Exposure to that light can then either activate or inhibit the nerve, which can give scientists information about how the nerve works and interacts with its surroundings.
Neuroscientists have applied optogenetics in animals to precisely trace the neural pathways underlying a range of brain disorders, including addiction, Parkinson’s disease, and mood and sleep disorders—information that has led to targeted therapies for these conditions.
To date, optogenetics has been primarily employed in the brain, an area that lacks pain receptors, which allows for the relatively painless implantation of rigid devices. However, the rigid devices can still damage neural tissues. The MIT team wondered whether the technique could be expanded to nerves outside the brain. Just as with the brain and spinal cord, nerves in the peripheral system can experience a range of impairment, including sciatica, motor neuron disease, and general numbness and pain.
Optogenetics could help neuroscientists identify specific causes of peripheral nerve conditions as well as test therapies to alleviate them. But the main hurdle to implementing the technique beyond the brain is motion. Peripheral nerves experience constant pushing and pulling from the surrounding muscles and tissues. If rigid silicon devices were used in the periphery, they would constrain an animal’s natural movement and potentially cause tissue damage.
Crystals and light
The researchers looked to develop an alternative that could work and move with the body. Their new design is a soft, stretchable, transparent fiber made from hydrogel—a rubbery, biocompatible mix of polymers and water, the ratio of which they tuned to create tiny, nanoscale crystals of polymers scattered throughout a more Jell-O-like solution.
The fiber embodies two layers—a core and an outer shell or “cladding.” The team mixed the solutions of each layer to generate a specific crystal arrangement. This arrangement gave each layer a specific, different refractive index, and together the layers kept any light traveling through the fiber from escaping or scattering away.
The team tested the optical fibers in mice whose nerves were genetically modified to respond to blue light that would excite neural activity or yellow light that would inhibit their activity.
They found that even with the implanted fiber in place, mice were able to run freely on a wheel. After two months of wheel exercises, amounting to some 30,000 cycles, the researchers found the fiber was still robust and resistant to fatigue, and could also transmit light efficiently to trigger muscle contraction.
The team then turned on a yellow laser and ran it through the implanted fiber. Using standard laboratory procedures for assessing pain inhibition, they observed that the mice were much less sensitive to pain than rodents that were not stimulated with light. The fibers were able to significantly inhibit sciatic pain in those light-stimulated mice.
The researchers see the fibers as a new tool that can help scientists identify the roots of pain and other peripheral nerve disorders.
“We are focusing on the fiber as a new neuroscience technology,” Liu says. “We hope to help dissect mechanisms underlying pain in the peripheral nervous system. With time, our technology may help identify novel mechanistic therapies for chronic pain and other debilitating conditions such as nerve degeneration or injury.”
More information:
Fatigue-resistant hydrogel optical fiber enables peripheral nerve optogenetics during locomotion, Nature Methods (2023). DOI: 10.1038/s41592-023-02020-9
Journal information:
Nature Methods
Source: Read Full Article